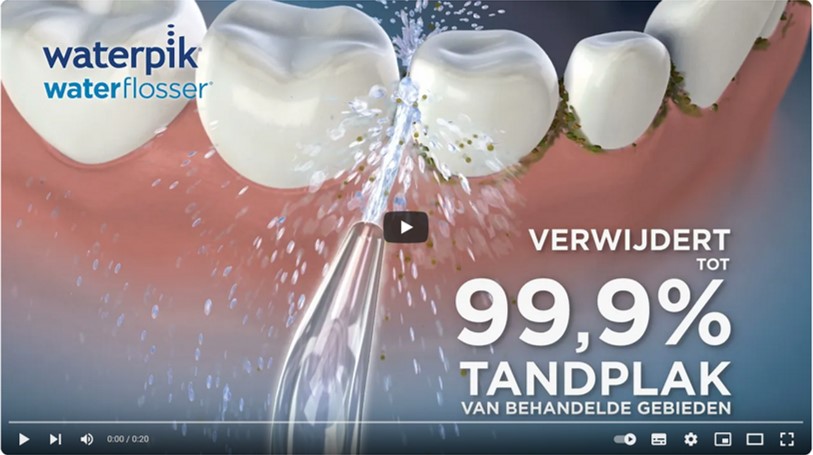
Wat doet een Waterpik® Waterflosser?
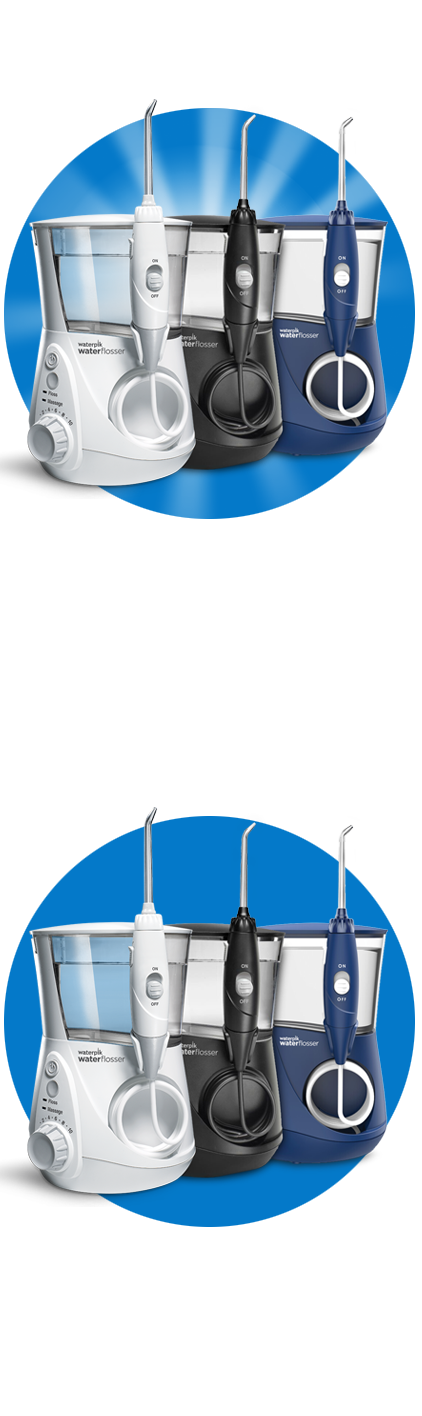
Ontdek Waterpik® Waterflossers
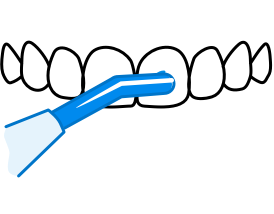
Uw mondgezondheid
Reiniging implantaten
Reinigen rondom implantaten, kronen of bruggen
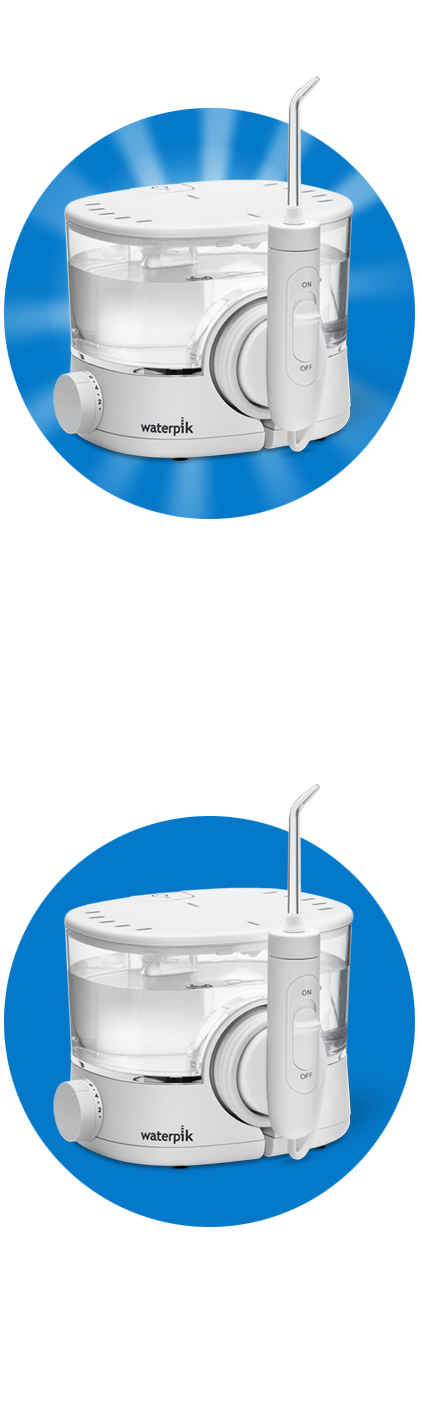
Natuurlijk wittere tanden
Op natuurlijke wijze uw tanden witter maken
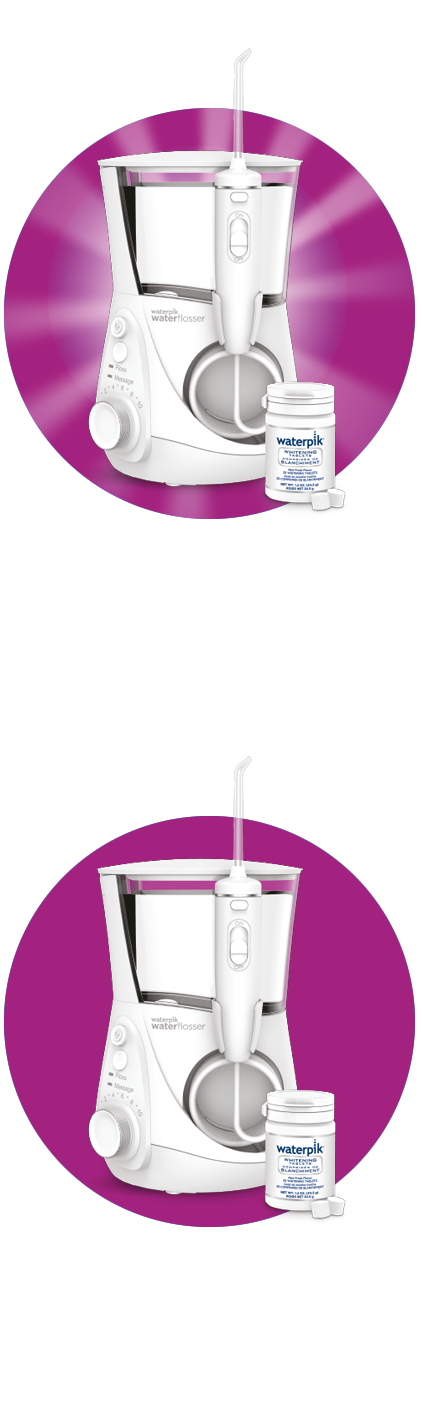
Populaire Waterpik Waterflossers
Waarom poetsen alleen niet genoeg is
De makkelijkste en meest effectieve manier om tussen de tanden te reinigen.
Alle Waterpik® Waterflossers zijn klinisch bewezen effectief.